

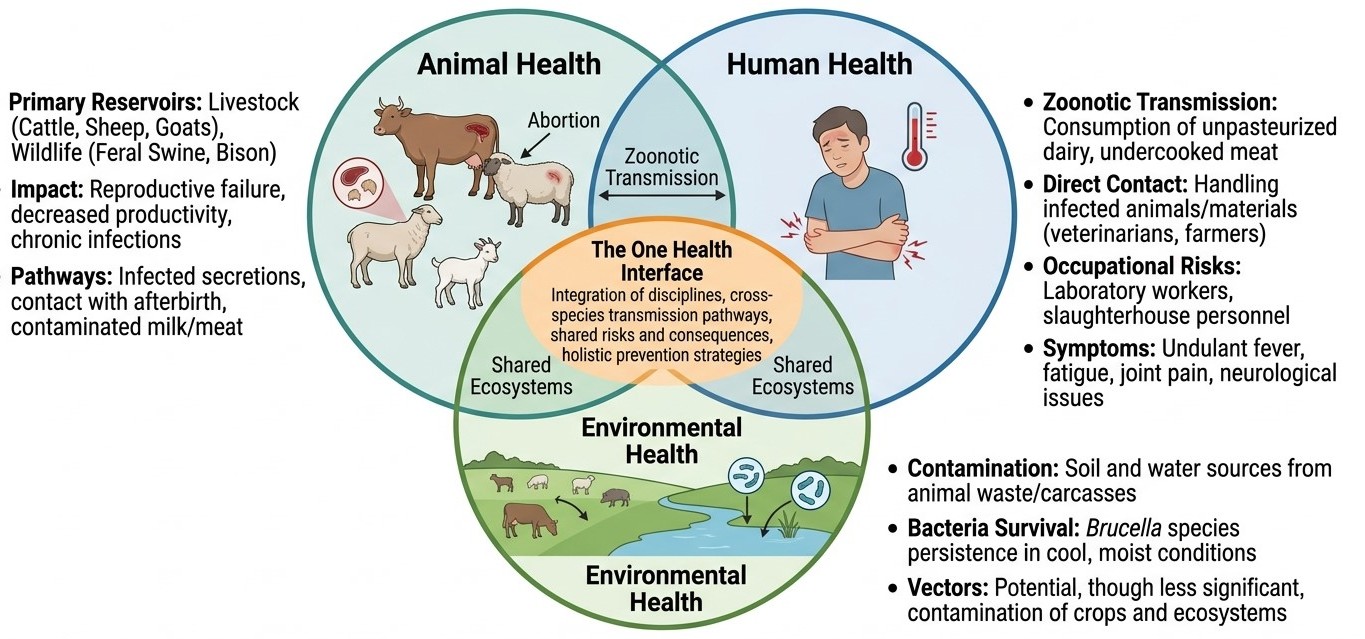
Brucellosis exemplifies the inseparable interconnection between animal health, human health, and environmental factors, making it an ideal candidate for application of the One Health framework. This comprehensive review examines the epidemiology, pathogenesis, transmission dynamics, clinical manifestations, diagnostic methodologies, treatment strategies, and prevention and control measures of human brucellosis through the lens of the One Health approach. The One Health concept recognizes that human health is inextricably linked to animal health and the health of the ecosystems in which they coexist. Brucellosis primarily affects livestock species, including cattle (Brucella abortus), sheep and goats (Brucella melitensis), swine (Brucella suis), and dogs (Brucella canis), with Brucella melitensis and Brucella abortus accounting for the majority of human infections worldwide. Transmission to humans occurs through direct contact with infected animals or their products, consumption of unpasteurized dairy products, and occupational exposure, making it a particular hazard for farmers, veterinarians, laboratory workers, and abattoir personnel. Effective control of human brucellosis requires coordinated veterinary, medical, and public health interventions. Vaccination of reservoir hosts, improved animal husbandry practices, pasteurization of dairy products, occupational safety measures, and robust surveillance systems form the pillars of a One Health-based control strategy. The review highlights significant gaps in integrated surveillance, transdisciplinary collaboration, and resource allocation in low- and middle-income endemic countries. Emerging challenges, including antimicrobial resistance, wildlife reservoirs, and climate-driven expansion of endemic zones, necessitate a holistic, ecosystem-based approach. Adopting the One Health paradigm in national and international brucellosis control programs offers the most promising pathway toward achieving sustained reduction in disease burden across species and ecosystems.
Shiga toxin-producing Escherichia coli (STEC) are significant zoonotic pathogens worldwide, causing diseases ranging from diarrhea to hemolytic uremic syndrome (HUS). Ruminants, mainly cattle, are considered the primary reservoirs; however, the role of horses in the epidemiology of STEC remains poorly understood in Nigeria. A total of 231 rectal swabs were collected from horses across selected equine facilities in five Nigerian states. Samples were processed following standard procedures, and isolates were confirmed using the Analytical Profile Index (API 20E) kit. Antibiotic susceptibility testing (AST) was performed using the Kirby-Bauer disk diffusion method with a panel of nine antibiotics, and the isolated Escherichia (E.) coli were classified as sensitive or resistant according to CLSI guidelines. The Multiple Antibiotic Resistance (MAR) index was calculated to assess the risk of contamination. The E. coli isolates were screened for selected Shiga toxin genes (stx1, stx2 and eae) using polymerase chain reaction (PCR). Fourteen isolates (6%) were identified as Shiga toxin-producing E. coli; of these, 78.6% (11/14) expressed at least one virulence gene: 42.9% (6/14) carried stx2, 35.7% (5/14) carried eae, and no isolate carried stx1. All confirmed STEC isolates exhibited multidrug resistance (MDR). The isolates showed complete resistance (100%) to cefuroxime and ciprofloxacin, while all were susceptible to ofloxacin. MAR indices ranged from 0.2 to 0.8, with 77.8% of isolates exhibiting MAR indices ≥0.5. This study reports the detection of MDR STEC in horses in Nigeria, revealing high-level resistance to clinically important antibiotics and significant carriage of virulence genes. The high MAR indices indicate high-risk contamination sources. The detection of MDR STEC in this study underscores a significant health risk to horses, with potential for zoonotic transmission to humans and the environment.
Not applicable
Not applicable
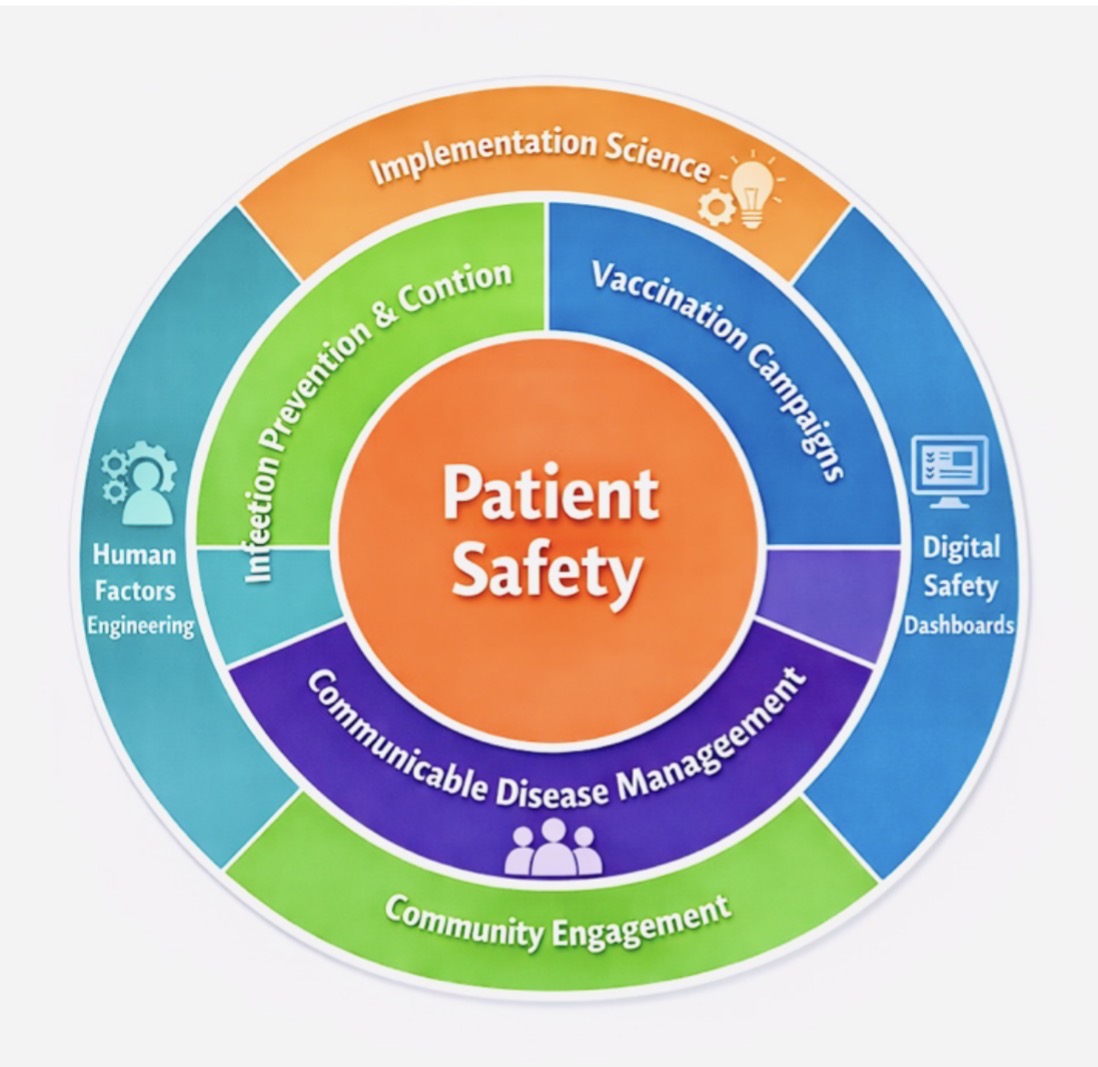
Patient safety is essential, and Infection Prevention and Control (IPC) has been shown to prevent harm. In primary care and outpatient settings, rising patient volumes, new care models, and emerging infectious threats require integrated safety systems. Aster Clinics in the United Arab Emirates (UAE) and Bahrain launched a comprehensive safety program that combined IPC, vaccination, and disease prevention strategies. The program was implemented from July 2025 to December 2025 across 80 clinics. Using a mixed-methods approach, key measures included standardized protocols, staff training, real-time audits, digital safety dashboards, vaccination outreach, and community campaigns. Quantitative data included IPC compliance, vaccination rates, and incident reports, while qualitative data assessed staff engagement, confidence, and safety culture. The program improved hand hygiene, vaccination rates, and transparency in incident reporting, fostering organizational learning. Digital tools enabled early detection and rapid response to safety issues. Staff gained confidence and participated more actively in safety discussions. Integrating IPC, vaccination, and disease management within a systems-based approach can enhance primary care safety, demonstrating how implementation science, human factors, digital tools, and community efforts strengthen resilient healthcare systems and promote a lasting safety culture.
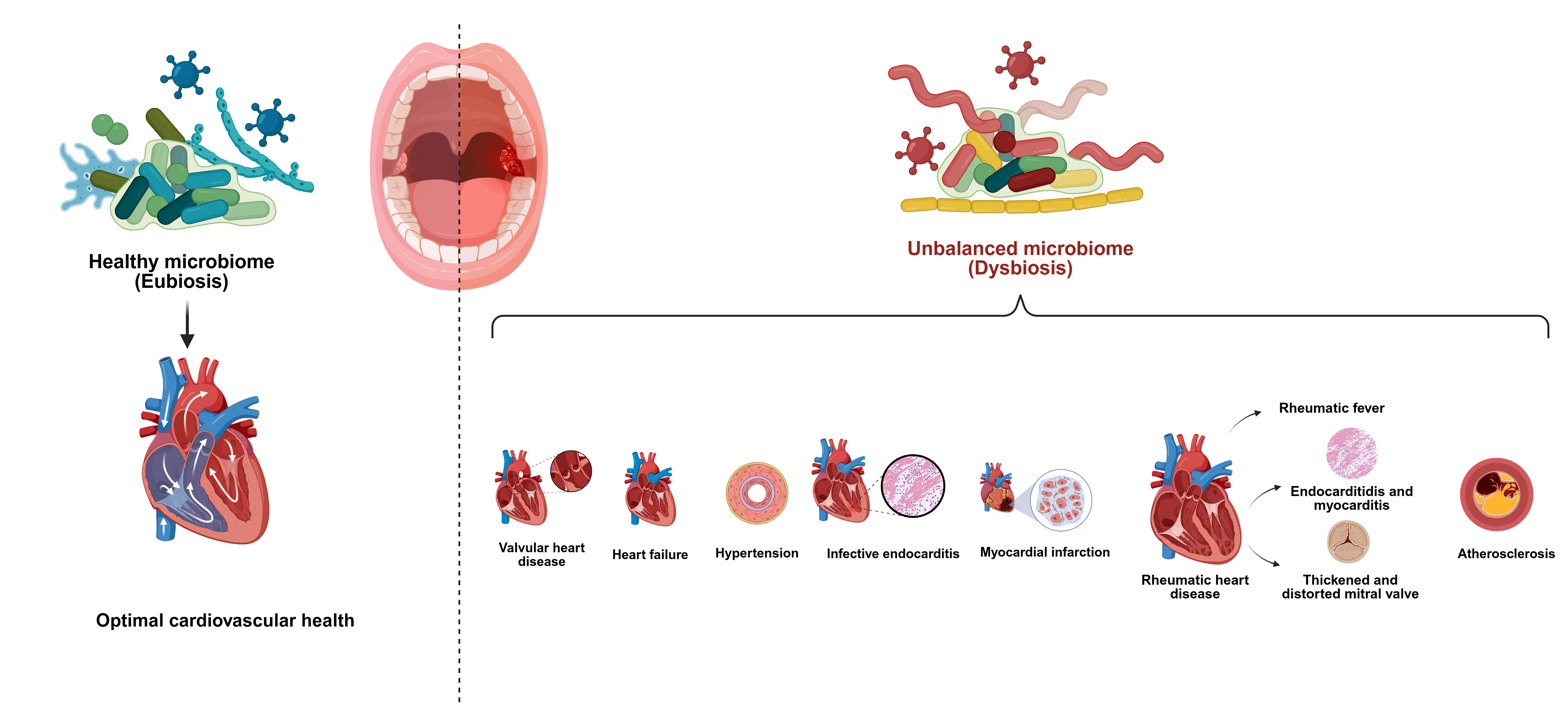
The oral microbiota begins to establish at birth and develops over time under the influence of genetic factors, environmental exposures, diet, and individual oral health behaviors. In a healthy state, these microorganisms exist in a balanced ecosystem; however, disruption of this balance, known as oral dysbiosis, can promote oral diseases such as dental caries and periodontitis (PD). Growing scientific evidence indicates that the impact of oral microbial imbalance may extend beyond the oral cavity. With advances in molecular detection techniques, oral bacterial species have been identified in distant body sites, including heart valves, suggesting a possible association between oral dysbiosis and cardiovascular disease. Chronic periodontal inflammation and microbial imbalance may allow bacteria or their inflammatory products to enter the bloodstream, leading to bacteremia via direct invasion or immune-mediated mechanisms. These processes may contribute to systemic inflammation and potentially influence the development and progression of cardiovascular disease. This study aims to review and summarize current evidence on the relationship between oral dysbiosis and cardiovascular disease, with particular focus on the mechanisms by which chronic oral inflammation and microbial imbalance may contribute to bacteremia and systemic inflammatory pathways that influence cardiovascular health.
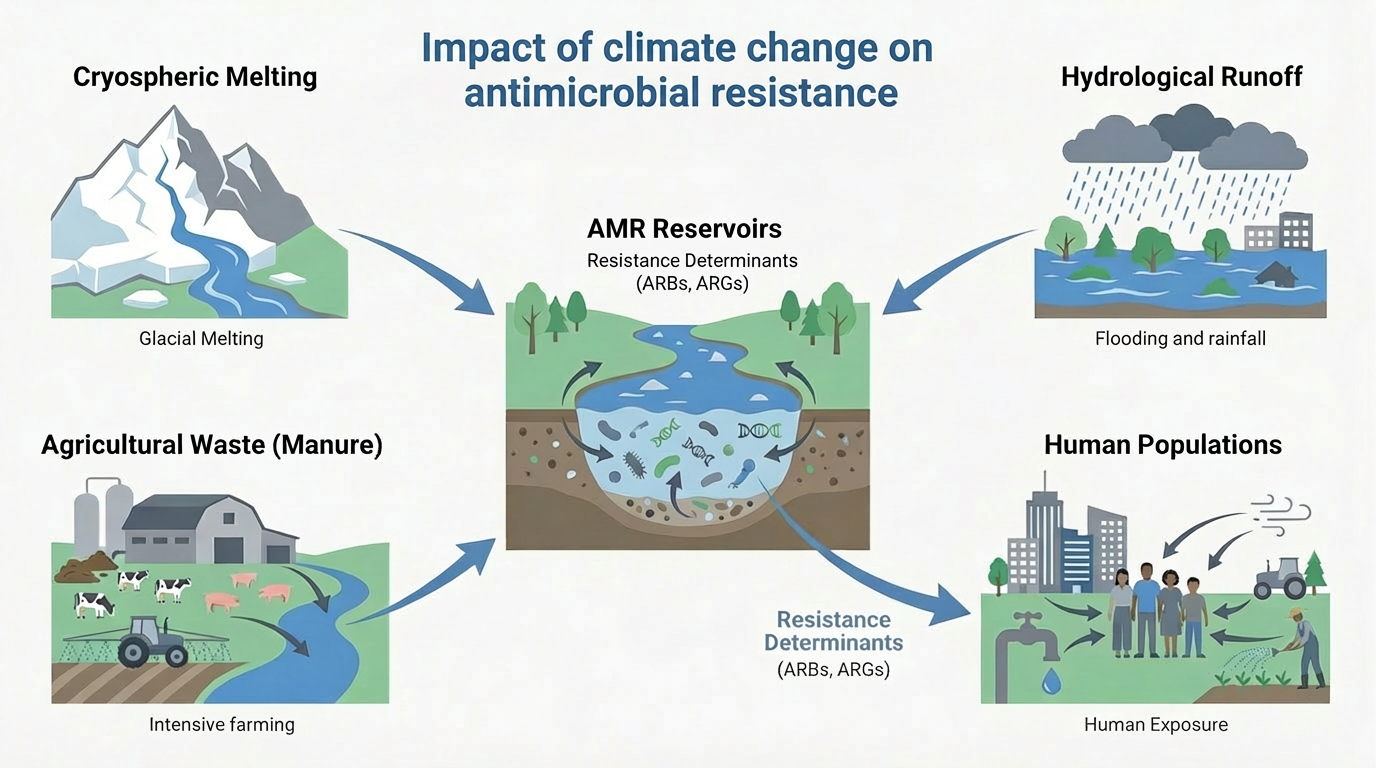
Antimicrobial resistance (AMR) and climate change are increasingly recognized as interconnected global threats driven by shared ecological, infrastructural, and socioeconomic vulnerabilities. This review summarizes emerging mechanistic and epidemiological evidence demonstrating how rising temperatures, hydrological extremes, ecosystem disruption, and environmental pollution accelerate the selection, persistence, and spread of antimicrobial resistance genes at the human-animal-environment interface. We emphasize climate-sensitive pathways, such as heat-driven microbial evolution, hydrological mobilization of resistance factors, aerosolized transmission, and climate-stressed food systems, which collectively reshape the distribution and impact of resistant bacterial infections. Importantly, we identify practical, co-beneficial interventions including pollution control, climate-resilient water and sanitation systems, adaptive antimicrobial stewardship, AI-powered surveillance, and integrated One Health governance. Framing AMR as a climate-sensitive health outcome shifts it from a mostly reactive, downstream clinical challenge to a predictable, preventable upstream systems vulnerability influenced by climate dynamics. Incorporating climate awareness into AMR policies and actions is therefore vital to preserving antimicrobial effectiveness, bolstering health system resilience, and safeguarding climate-sensitive populations in a rapidly warming world.
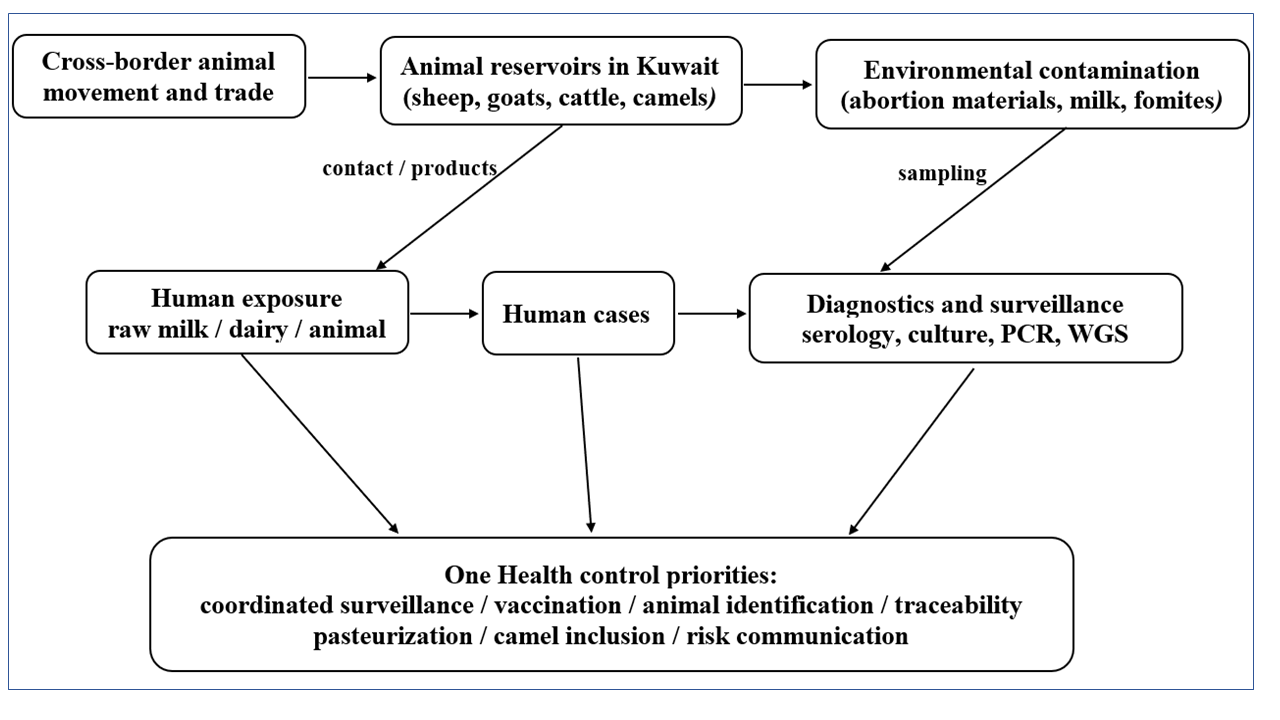
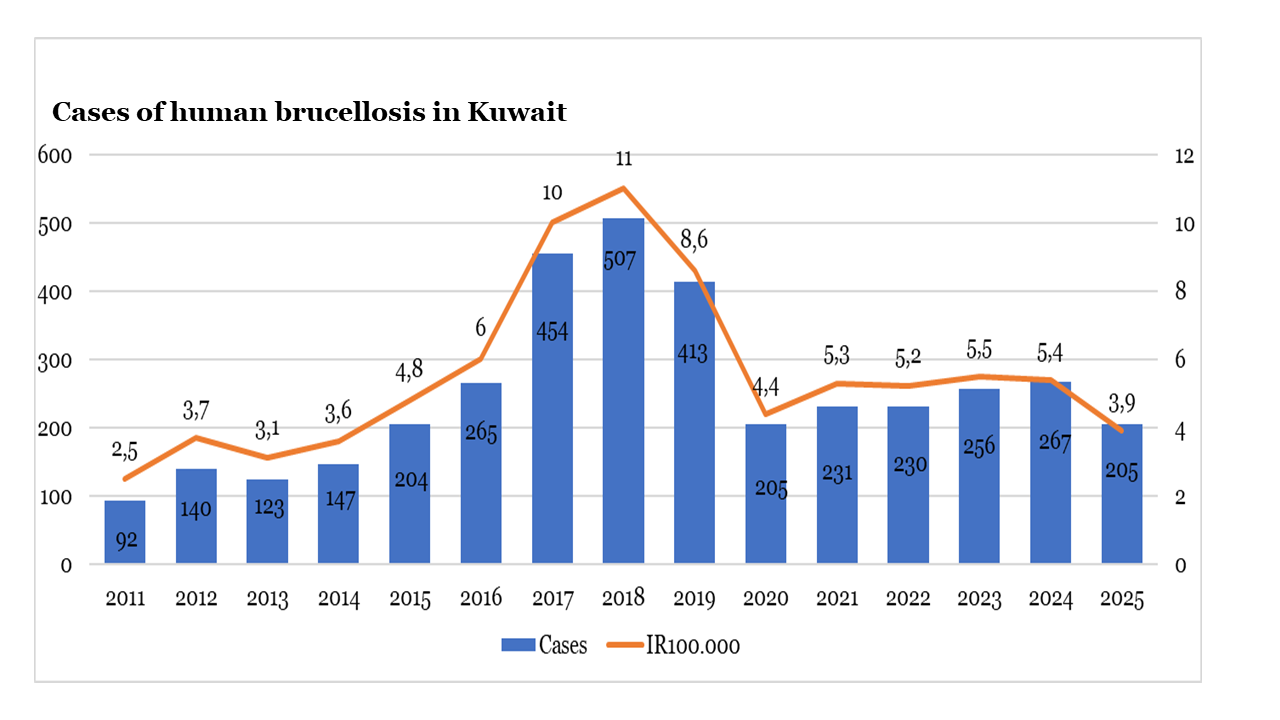
Brucellosis remains one of the most important endemic zoonoses in Kuwait, affecting both human and animal populations despite decades of control efforts. This review synthesizes published literature and official data on the epidemiology of brucellosis in Kuwait from a One Health perspective, integrating evidence from human health, veterinary surveillance, and livestock control programs. Historical data show a substantial burden of human disease during the 1980s and 1990s, characterised by frequent outbreaks and severe complications, followed by fluctuating incidence in subsequent decades. Animal brucellosis remains endemic, particularly among sheep, goats, cattle, and camels, with Brucella melitensis identified as the predominant species in both humans and livestock. Vaccination programs have reduced prevalence but have not achieved sustained elimination. Persistent transmission is driven by close human–animal contact, consumption of unpasteurized dairy products, and gaps in coordinated surveillance and control. The findings highlight the need for integrated One Health strategies, strengthened surveillance, and sustained livestock vaccination to reduce the ongoing public health and economic impact of brucellosis in Kuwait.
Listeriosis is a significant zoonotic disease affecting ruminants and causing substantial economic losses in livestock production. Although numerous regional studies have been conducted in Türkiye, a comprehensive synthesis of available data has been lacking. The current study aimed to evaluate the epidemiology, clinical forms, diagnostic approaches, and regional distribution of animal listeriosis in Türkiye. Studies reporting clinical listeriosis in animals in Türkiye between 1980 and 2024 were included. Due to methodological heterogeneity, a descriptive-analytic approach was adopted. A total of 26 studies were included. Most studies were conducted in Eastern and Central Anatolia, regions with intensive ruminant farming and widespread silage use. Sample sizes ranged from 18 to 3,760 animals. The most frequently investigated species were sheep (n=13 studies) and cattle (n=9 studies), followed by goats, dogs, and horses. Molecular and culture-based studies generally reported low positivity rates (0–7.29%), whereas serological studies showed a substantially wider range (17–86.69%), suggesting past exposure rather than active infection. An exceptionally high culture positivity rate (66.6%) was reported in a small-scale investigation of an encephalitic outbreak, whereas 100% seropositivity was observed in limited farm-based screenings of horses and dogs. Both findings likely reflect context-specific findings from small samples rather than population-level prevalence. Clinically, encephalitic and abortive forms were predominant in small ruminants, whereas subclinical infections were frequently reported in cattle through serological surveillance. The available evidence indicates that contaminated silage is a major epidemiological risk factor, particularly in regions with prolonged winter feeding. Notably, national surveillance data remain scarce, and most studies were cross-sectional or case-based. In conclusion, animal listeriosis remains a significant health concern in Türkiye, particularly in ruminant-dense regions. Strengthening surveillance and expanding molecular diagnostics in primary animal production farms, along with integration within a One Health framework, are essential to better define the disease burden and reduce zoonotic risk.
Carbapenem‑resistant Enterobacteriaceae (CRE) are associated with high morbidity and mortality in humans due to very limited therapeutic options. This systematic review examines the prevalence and geographic distribution of CRE, as well as the associated genetic factors at the human-animal-environment interface in Africa. To extract information for this study, the Medical Subject Headings (MeSH) technique was used to identify studies that employed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines, with a focus on original research articles. Additionally, this systematic review employed the CoCoPop (conditions, context, and population) framework to identify relevant articles. Search engines used to retrieve articles included Web of Science, Google Scholar, Scopus, ResearchGate, and PubMed. By August 2025, only eight articles from Africa met the search criteria and were included in the analysis. The observed prevalence of CRE across the heterogeneous studies was 18.58%. The animal domain exhibited the highest CRE prevalence (9.89%), followed by the human (6.06%) and environmental (2.63%) domains. Escherichia coli (58.35%) and Klebsiella pneumoniae (28.28%) were the predominant spp. among the reported CRE isolates. The highest gene prevalence was observed for blaOXA-48 (42%), followed by blaKPC (33%) and blaOXA-66 (9.78%). Geographically, North Africa had the highest prevalence (55.78%), followed by Eastern Africa (29.82%), whereas Southern Africa had the lowest prevalence (3.08%). Our study emphasizes the significant burden of CRE infections and genes, as well as their spread across human, animal, and environmental sectors in Africa. The simultaneous presence of CRE in these areas indicates potential interfaces, but current evidence does not permit a definitive assignment of specific transmission routes. Surveillance programs, research, infection prevention, and control measures from a One Health perspective should be implemented to inform interventions.
The One Health approach has emerged as a critical framework for addressing zoonotic diseases and infections during outbreaks worldwide. By recognizing the interconnectedness of human, animal, and environmental health, One Health promotes cross-sector collaboration essential to early detection, prevention, and response. However, translating this concept into practice requires more than policy endorsement; it demands operational integration, shared data platforms, and equitable partnerships that prioritize local leadership. From this point, we are delighted to announce the launch of the One Health Microbiology & Infection (One Health Microbiol. Infect.) journal, a dedicated platform for advancing research, innovation, and knowledge dissemination in the rapidly evolving fields of microbiology and infection within One Health insights.
